Date: 3rd February 2022
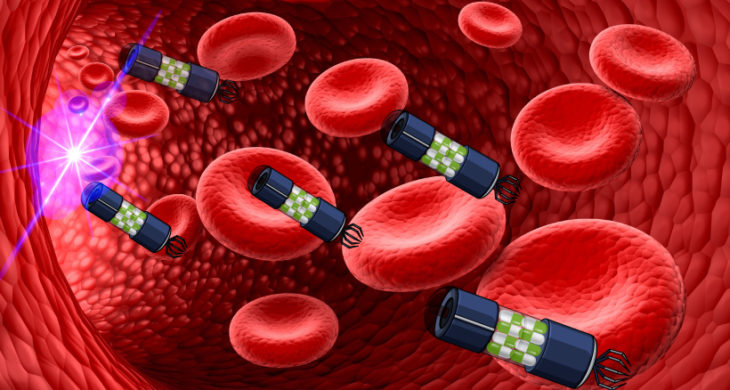
Drug delivery systems are engineered technologies for the targeted delivery and/or controlled release of therapeutic agents. They can in principle provide enhanced efficacy, reduced toxicity and can lower an agents chance of triggering drug resistance. Medicine are undergoing a revolution, and the next generation of therapies are different from traditional ones, and require advancements in delivery systems. Now, researchers have developed organic microparticles, or microswimmers, that can steer through biological fluids and blood, propelled at high speed by light and deliver cargo.
Microswimmers represent a class of micromachines that are able to convert external energy to swimming motion. Microswimmers have opened up avenues for diverse biomedical applications, including targeted drug delivery , precision nanosurgery, and diagnostic sensing. In particular, light-driven microswimmers show distinct advantages, as the energy source of propulsion is convenient and allows for high spatial and temporal remote control. However, a major challenge for biomedical application for light-driven microswimmers has been their intolerance to salts found in water or the body.
Now, researchers at the Max Planck Institute for Intelligent Systems and the Max Planck Institute for Solid State Research, Germany, led by Metin Sitti and Bettina Lotsch, have developed on the spot drug delivery with light-controlled organic microswimmers. The microparticles are propelled at high speed by visible light, either individually or as a swarm. Additionally, they are partially biocompatible and can take up and release cargo on demand.
The scientists used a porous two-dimensional carbon nitride (CNx) that could be synthesised from organic materials, for instance, urea. Similar to solar panels, carbon nitride can absorb light which then provided the energy to propel the robot forward when light illuminated the particle surface. The porous structure of the nanoparticles allows ions to flow through them, which reduced the resistance created by the salt and allowed them to move rapidly through all biological fluids. The team demonstrated the microswimmers propelled at high-speed, 15-23 µm per second – equating to > 9 body lengths per second, and could swim in multicomponent ionic solutions with concentrations up to 5 M.
Next, the team assessed the ability of the microswimmers to carry and release drug cargo. The pores on the nanoparticle were loaded with the cancer drug, doxorubicin (DOX), and resulted in a high loading efficiency of 185% without passive release, and was stable for over a month. Controlled drug release was reported under different pH conditions and could be induced on-demand by illumination. Furthermore, the speed of the microswimmers when loaded was not compromised.
So how does the light, which enables propulsion, not simultaneously release the cargo in undesired locations? Here, the new carbon nitride had intrinsic charging, so when illuminated in an hypoxic environment, the microswimmer can charge up like a solar battery, this modifies the interaction with the adsorbed drug, and the drug release is significantly boosted. In effect, the material’s light charging ability, which is conditioned by hypoxic conditions, suddenly becomes a sensing property for the release. This is particularly relevant for targeting hypoxic regions such as oxygen-deprived tumours.
To prove this interaction the team illuminated DOX-loaded microswimmers in the vicinity of cancer cells, the drug was released and taken up by the cells, which resulted in their decay.
Conclusions and future applications
The team here have demonstrated that organic microswimmers can steer through biological fluids and dissolved blood in an unprecedented way and enable controlled on-demand cargo release. They overcome the current challenges to light-driven propulsion as they have a high ion tolerance, and have fuel-free high speed propulsion.
The team recognise that microswimmer technology for biomedical applications is a vision of the future. However, this research lays down the foundations towards light-controlled and biocompatible material, as well as intelligent semi-autonomous systems. Currently these microbots only work in the most optimal conditions, so broadening these limitations, enhancing cargo uptake, or further tailoring of molecular site could enable more controlled cargo interactions in the future.
The work here should add to the growing use of nanoparticles for biomedical purposes. We have recently seen tolerogenic ImmTOR nanoparticles enhance hepatic gene therapy, ligand-bound nanoparticles to treat osteoarthritis, and even nanoparticles to treat genetic diseases in utero. However, each have their own characteristics and nuances, and advancing light-driven delivery vehicles will be an valuable addition to the delivery system toolbox.
For more information please see the press release at the Max Planck Institute for Intelligent Systems
Sridhar, V., Podjaski, F., Alapan, Y., Kröger, J., Grunenberg, L., Kishore, V., Lotsch, B.V., and Sitti, M. (2022). Light-driven carbon nitride microswimmers with propulsion in biological and ionic media and responsive on-demand drug delivery. Science Robotics 7, eabm1421.
https://doi.org/10.1126/scirobotics.abm1421
http://eha.3a7.mywebsitetransfer.com/ligand-bound-nanoparticles-treat-osteoarthritis/
http://eha.3a7.mywebsitetransfer.com/nanoparticles-treat-genetic-diseases-in-utero/
http://eha.3a7.mywebsitetransfer.com/tolerogenic-immtor-nanoparticles-enhance-hepatic-gene-therapy/